Atoms Test Study Stacks
Information to review for Atom Test
Flashcards http://www.studystack.com/inewflashcard-688187
Matching http://www.studystack.com/istudymatch-688187
Hangman http://www.studystack.com/ihangman-688187
Bug Match http://www.studystack.com/ibugmatch-688187
Unscramble http://www.studystack.com/iwordscramble-688187
Quiz to Practice (this is NOT the real test) http://www.studystack.com/iquiz-688187
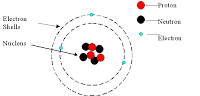
Know how to look at this and tell how many:
Protons
Neutrons
Electrons
are in the atom (an example of a Lithium Atom is below you can download or view the attachment)
Know how to use information from the periodic table to find out the following information.
Atomic # _____________
Atomic Mass _____________
# of Protons _______________
# of Neutrons ______________
# of Electrons _______________
Draw a Nitrogen Atom. Label (+) charged particle, (-) charged particle, and (0) neutral charged particle. Color the proton yellow, neutron orange, and electron grey.
Information to review for Atom Test
Flashcards http://www.studystack.com/inewflashcard-688187
Matching http://www.studystack.com/istudymatch-688187
Hangman http://www.studystack.com/ihangman-688187
Bug Match http://www.studystack.com/ibugmatch-688187
Unscramble http://www.studystack.com/iwordscramble-688187
Quiz to Practice (this is NOT the real test) http://www.studystack.com/iquiz-688187
Know how to look at this and tell how many:
Protons
Neutrons
Electrons
are in the atom (an example of a Lithium Atom is below you can download or view the attachment)
Know how to use information from the periodic table to find out the following information.
Atomic # _____________
Atomic Mass _____________
# of Protons _______________
# of Neutrons ______________
# of Electrons _______________
Draw a Nitrogen Atom. Label (+) charged particle, (-) charged particle, and (0) neutral charged particle. Color the proton yellow, neutron orange, and electron grey.
